Uveitis – Canine
Rachel L. (Mathes) Davis1
Veterinary Information Network (VIN)
Abstract
Uveitis is a general term referring to inflammation of the vascular tunic of the eye, encompassing the iris, ciliary body and choroid.1 The term uveitis is typically used to reference anterior uveitis or inflammation of the iris (iritis) and ciliary body (cyclitis). The ciliary body component is clinically presumed as cyclitis is typically a histopathologic diagnosis due to the location of the ciliary body in the eye. Thus clinically, uveitis is usually described based on changes to the iris and other visible anterior segment structures in the eye. Although uveitis technically encompasses inflammation of all vascular tunics, more descriptive terms are typically used when referencing posterior segment or complete ocular inflammation.2 Choriodal (+/- retinal) inflammation, termed choroiditis or chorioretinitis, may occur independently of or concurrent with anterior uveitis, depending on the underlying cause. Endophthalmitis refers to inflammation of the iris, ciliary body and choroid with concurrent pathology to surrounding intraocular structures. Panuveitis refers to inflammation of the vascular tunic as well as entire globe and may result in signs of orbital disease depending on the severity of disease
Uveitis in dogs is inflammation of the uveal tract, including the iris, ciliary body, and choroid. Learn about causes, diagnosis, ophthalmic findings, treatment options, systemic evaluation, and prognosis for canine uveitis below.
Keywords: uveitis, canine uveitis, anterior uveitis dogs, posterior uveitis dogs, panuveitis dogs, endophthalmitis dogs, chorioretinitis dogs, iritis dogs, cyclitis dogs, dog eye inflammation, ocular inflammation dogs, intraocular inflammation dogs, secondary glaucoma dogs, canine ocular disease, ocular ultrasound dogs, anterior chamber flare dogs, keratic precipitates dogs, aqueous flare dogs, prednisolone acetate dogs, atropine dogs, veterinary ophthalmology
Contributors:
1Revised by Rachel Davis DVM, MS, DACVO at Animal Eye Clinic, Westfield, Indiana, USA, October 2016
Correspondence:
Rachel L. Davis, DVM, MS, Diplomate, ACVO – Ophthalmologist
Animal Eye Clinic
4750 Killarney Drive
Carmel, IN 46033
Email: info@indyaec.com
Disease Description
Uveitis is a general term referring to inflammation of the vascular tunic of the eye, encompassing the iris, ciliary body and choroid.1 The term uveitis is typically used to reference anterior uveitis or inflammation of the iris (iritis) and ciliary body (cyclitis). The ciliary body component is clinically presumed as cyclitis is typically a histopathologic diagnosis due to the location of the ciliary body in the eye. Thus clinically, uveitis is usually described based on changes to the iris and other visible anterior segment structures in the eye. Although uveitis technically encompasses inflammation of all vascular tunics, more descriptive terms are typically used when referencing posterior segment or complete ocular inflammation.2 Choriodal (+/- retinal) inflammation, termed choroiditis or chorioretinitis, may occur independently of or concurrent with anterior uveitis, depending on the underlying cause. Endophthalmitis refers to inflammation of the iris, ciliary body and choroid with concurrent pathology to surrounding intraocular structures. Panuveitis refers to inflammation of the vascular tunic as well as entire globe and may result in signs of orbital disease depending on the severity of disease.
Etiology
Uveitis is a general term that does not denote any specific underlying etiology.3 The causes of uveitis are numerous and, in part, depend on the dog’s geographic location, travel history, environment, age, sex and breed. Several grouping categories have been proposed to help further classify underlying causes of uveitis. Etiologies may be classified based on endogenous or systemic causes (e.g. infectious, neoplastic) and exogenous or external causes (e.g. toxic, traumatic, radiation).2 Typically, exogenous causes can be readily identified (e.g. penetrating ocular injury, accidental bleach or cleaning product contact, etc.). While unwitnessed trauma is often an attractive “explanation” for the owner and practitioner, this is an uncommon to rare cause of uveitis, especially if there are no other periocular or systemic signs of trauma. Endogenous causes of uveitis are far more common than exogenous and, as such, can be difficult to elucidate. Endogenous causes can be further categorized based on primary ocular disease (e.g. lens-induced uveitis, intraocular neoplasia, pigmentary uveitis) versus systemic disease (e.g. infectious, neoplastic, immune-mediated, toxic, metabolic, idiopathic). Again, intraocular causes are typically readily identified, although diagnostics such as ocular ultrasound or histopathology may be required for definitive diagnosis. Systemic causes of uveitis are the most common underlying etiology, which may present a diagnostic challenge for the practitioner.3 Anterior uveitis is the leading cause of secondary glaucoma in dogs4,5 and is a vision-threatening and often globe-threatening condition, therefore, systemic work up is warranted for most patients with uveitis.
In addition to the extensive list of known causes of uveitis in dogs (insert previous references), more recently, Aspergillus sp.,6 Leishmaniasis,7,8 Candida sp.,9 pine processionary caterpillar setae,10 E. cuniculi,11 Brucella sp.,12 Onchocerca lupi,13 pit viper bites,14 ocular alkaline injury,15 Sarcocystis neurona,16 mastocytosis,17 Trypanosoma evansi,18 septic implantation syndrome,19 toxocariasis,20 uveodermatologic lymphoma,21 leptospirosis,22 corneal and anterior segment foreign body trauma23 and necrotizing scleritis24 have been identified or further characterized as causes of canine uveitis.
Diagnosis
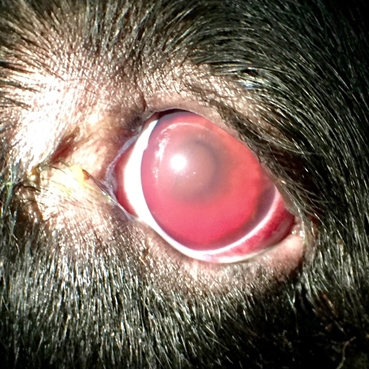
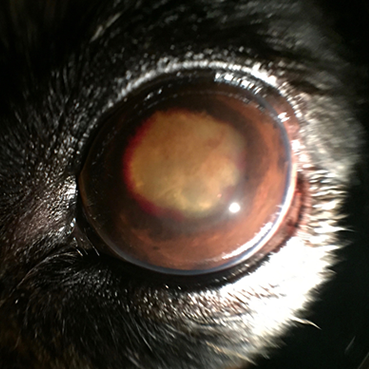
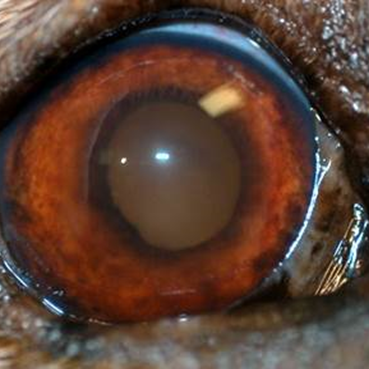
Ophthalmic Examination Findings: The ophthalmic findings may vary considerably depending on the duration, severity and nature of the uveitis. Consistent findings of anterior uveitis include rubeosis, iridal swelling, “sluggish” or absent iris movement and flare, the hallmark of uveitis. Miosis is typically also observed, although mydriasis may be present if secondary glaucoma is present. Sluggish iris movement may be noted as slow or minimal PLR responses or may also be seen as incomplete pharmacologic mydriasis (dilation) if topical atropine or tropicamide is instilled. Flare, caused by the Tyndall effect,25 is due to increased cell content of the aqueous from leakage of the iridal vessels and is assessed by using the slit beam aperture on a focal light source and viewing the anterior chamber from the side (or holding the light source to the side and viewing the eye centrally). Depending on the severity of the anterior uveitis, hyphema, hypopyon, intraocular fibrin, pre-iridal fibrovascular membranes, cataracts, iridal darkening, corneal edema, keratic preciptates and/or obscured visibility of intraocular structures may be present. Choroiditis or chorioretintis may be seen as intraretinal edema, hyporeflectivity or absence of the tapetal structures, granulomas, retinal detachment, yellow to tan intraretinal or intrachoroidal infiltrates. If panuveitis or endophthalmitis is present, posterior segment pathology may be obscured by anterior changes and may not be fully recognized. Depending on the severity of the uveitis, vision may be decreased or absent.
Non-specific ophthalmic findings may also include blepharospasm, photophobia, conjunctival hyperemia, scleral injection, ciliary flush and enophthalmos with third eyelid elevation.1
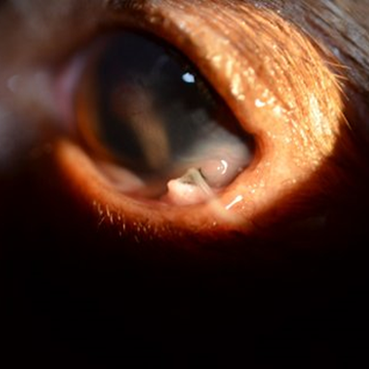
Physical Examination Findings: Unless there is an obvious exogenous underlying cause (Figure 2), most cases of canine uveitis present without other overt clinical abnormalities. Because most cases of canine uveitis are caused by systemic disease, a full physical examination and detailed clinical history are important when assessing these patients. Typically; however, further diagnostics are required beyond a physical examination to assess for underlying causes of uveitis as many affected patients are otherwise healthy.26
Disease Description in this Species
Signalment
Uveitis may affect any age, breed or sex of dog.
Clinical Signs
Ophthalmic findings: rubeosis, iridal swelling, flare, sluggish iridal movement, miosis (unless secondary glaucoma present), hyphema, hypopyon, keratic precipitates, intraocular fibrin, loss of iridal detail, pre-iridal fibrovascular membranes, cataracts, iridal darkening, corneal edema, intraretinal edema, hyporeflective tapetum, loss of tapetal structures or detail, subretinal granulomas, retinal detachments, choroidal tan to yellow infiltrates, tan to yellow vitreal aggregates or “haze,” blepharospasm, conjunctival hyperemia, scleral injection, ciliary flush, enophthalmos, decreased vision
Clinical signs: highly varied, may be otherwise normal, cutaneous lesions, draining tracts, lameness, weight loss, anorexia, malaise, tachypnea, abdominal pain, abdominal or thoracic masses, hematologic abnormalities, many others
Etiology
Exogenous
- Penetrating injury
- Blunt trauma
- Toxin contact exposure
- Radiation
Endogenous (add previous list and list above)
Breed Predilection
- None
Sex Predilection
- None
Age Predilection
- None/any
Diagnostic Procedures
Ocular ultrasound: Cases of severe anterior segment pathology in which deeper intraocular structures are obscured may be evaluated using ocular ultrasound. This modality may help to further characterize intraocular pathology and assess for intraocular neoplasia or trauma.
Aqueocentesis: Anterior chamber aspiration (aqueocentesis) has not been shown to be a useful diagnostic tool when assessing uveitis in most cases. One study showed no benefit for canine cases27 while another study suggested that it may be helpful when diagnosing canine lymphoma.28 However, canine lymphoma is typically readily diagnosed using other systemic imaging and sampling modalities. Because aqueocentesis is of questionable value in canine cases of uveitis, requires general anesthesia and carries risks of worsening uveitis, lens trauma, intraocular bleeding or iris trauma, it is not recommended for use in cases of canine uveitis.
Ophthalmic examination: Because many changes present with anterior uveitis or chorioretinitis are difficult to characterize and often require magnification and advanced training for full evaluation, referral to a veterinary ophthalmologist may be warranted for patients affected with uveitis.
Systemic diagnostics: If exogenous causes and endogenous primary ocular causes are ruled out, systemic diagnostics are recommended in cases of canine uveitis. A baseline CBC and chemistry may be invaluable for assessing overall health. In many cases, this diagnostic is normal and further infectious disease testing (e.g. tick titers, toxoplasmosis titers, fungal antigen testing, etc.) and imaging (e.g. thoracic radiographs, abdominal radiographs, abdominal ultrasound, etc.) is warranted. Other diagnostics such as lymph node aspirates, skin cytology +/- biopsy of integument lesions or fine needle aspirates of other masses or lesions may also be warranted. In some cases, an underlying cause is not identified (idiopathic) or autoimmune disease is diagnosed based on exclusion of other causes.
Images
Treatment/Management/Prognosis
Specific Therapy
Specific therapy depends on identification of the underlying cause of uveitis. Topical anti-inflammatory steroidal (e.g. prednisolone acetate, dexamethasone) and non-steroidal (e.g. diclofenac, flurbiprofen) medications may be used in conjunction 2-4x per day depending on the severity of the uveitis.2,3 Oral anti-inflammatory therapy using steroids or NSAIDs may be pursued once the underlying cause of uveitis is identified. Oral steroids may worsen infectious disease if concurrent systemic therapy is not instituted and many NSAIDs are not effective at treating intraocular inflammation.2,29,30 Thus, their use should be judiciously pursued based on systemic evaluation. Because bacterial infections other than tick-borne diseases are uncommon to rare causes of uveitis, broad spectrum antibiotics are of no value in treating canine uveitis. Referral to a veterinary ophthalmologist and/or internist should be considered for many cases of canine uveitis.
Supportive Therapy
Topical cycloplegics (e.g. atropine) may be used to help prevent posterior synechia, provide analgesia and decrease ciliary spasm; however, their use should be judicious and patients on these medications should be closely monitored as cycloplegics may potentiate or worsen secondary glaucoma.2 Oral immune modulating therapy (e.g. azathioprine, cyclosporine) may be useful for cases of idiopathic or autoimmune uveitis; however, infectious and neoplastic causes of uveitis should be ruled out prior to their use. Subconjunctival steroids (e.g. triamcinolone) may help decrease intraocular inflammation but may cause subconjunctival granuloma formation or potentiate corneal infection or malacia if an ulcer is present or occurs.31
Monitoring and Prognosis
Patients with uveitis are at risk for vision or globe threatening complications, thus close monitoring, identification of underlying cause/s, prompt systemic work up and/or referral to an ophthalmologist are warranted when diagnosing uveitis. The prognosis is dependent on the underlying cause of uveitis. Client education regarding the severity of the disease and need for identification of underlying pathology is also important. Monitoring for secondary glaucoma with frequent intraocular pressure evaluation (every 1-4 weeks depending on the severity and nature of the uveitis) should also be recommended.
Differential Diagnosis
Other ocular conditions causing corneal edema, conjunctival hyperemia or scleral injection
References
- Townsend WM. Canine and feline uveitis. Vet Clin North Am Small Anim Pract. 2008 Mar;38(2):323-46.
- Hendrix DVH. Diseases and Surgery of the Canine Anterior Uvea. In Gelatt KN (ed): Veterinary Ophthalmology 4th ed. Pg 818-36. Blackwell Publishing, Ames IA
- van der Woerdt A. Management of intraocular inflammatory disease. Clin Tech Small Anim Pract. 2001 Feb;16(1):58-61.
- Johnsen DA, Maggs DJ, Kass PH. Evaluation of risk factors for development of secondary glaucoma in dogs: 156 cases (1999-2004). J Am Vet Med Assoc. 2006;15:1270-4.
- Strom AR, Hassig M, Iburg TM, et al. Epidemiology of canine glaucoma presented to the University of Zurich from 1995 to 2009. Part 2: secondary glaucoma (217 cases). Vet Ophthalmol. 2011;14:127-32.
- Wooff PJ, Dees DD, Teixeria L. Aspergillus spp. panophthalmitis with intralenticular invasion in dogs: report of two cases. Vet Ophthalmol. 2016 Sep 19.
- Koutinas AF, Koutinas CK. Pathologic mechanisms underlying the clinical findings in canine leishmaniasis due to Leishmania infantum/chagasi. Vet Pathol. 2014 Mar;51(2):527-38.
- Pietro SD, Bosco VR, Crinò C, et al. Prevalence, type, and prognosis of ocular lesions in shelter and owned-client dogs naturally infected by Leishmania infantum. Vet World. 2016 Jun;9(6):633-7.
- Enders A, van der Woerdt A, Donovan T. Endogenous mycotic endophthalmitis in a dog with candiduria and Evans syndrome. Vet Ophthalmol. 2016 Mar 3. epub ahead of print.
- Costa D, Esteban J, Sanz F, et al. Ocular lesions produced by pine processionary caterpillar setae (Thaumetopoea pityocampa) in dogs: a descriptive study. Vet Ophthalmol. 2015 Nov 26, epub ahead of print.
- Nell B, Csokai J, Fuchs-Baumgartinger A, et al. Encephalitozoon cuniculi causes focal anterior cataract and uveitis in dogs. Tierarztl Prax Ausg K Kleintiere Heimtiere. 2015;43(5):337-44.
- Ledbetter EC, Landry MP, Stokol T, et al. Brucella canis endophthalmitis in 3 dogs: clinical features, diagnosis, and treatment. Vet Ophthalmol. 2009 May-Jun;12(3):183-91.
- Komnenou AT, Thomas AL, Papadopoulos E, et al. Intraocular localization of Onchocerca lupi adult worm in a dog with anterior uveitis: A case report. Vet Ophthalmol. 2016 May;19(3):245-249.
- Martins BC, Plummer CE, Gelatt KN, et al. Ophthalmic abnormalities secondary to periocular or ocular snakebite (pit vipers) in dogs–11 cases (2012-2014). Vet Ophthalmol. 2016 Mar;19(2):149-60.
- Busse C, Hartley C, Kafarnik C, Pivetta M. Ocular alkaline injury in four dogs – presentation, treatment, and follow-up – a case series. Vet Ophthalmol. 2015 Mar;18(2):127-34.
- Dubey JP, Black SS, Verma SK, et al. Sarcocystis neurona schizonts-associated encephalitis, chorioretinitis, and myositis in a two-month-old dog simulating toxoplasmosis, and presence of mature sarcocysts in muscles. Vet Parasitol. 2014 May 28;202(3-4):194-200.
- Boostrom BO, Good KL, Maggs DJ, et al. Unilateral intraocular mastocytosis and anterior uveitis in a dog with subcutaneous mast cell tumors. Vet Ophthalmol. 2014 Mar;17(2):131-8.
- Defontis M, Richartz J, Engelmann N, et al. Canine Trypanosoma evansi infection introduced into Germany. Vet Clin Pathol. 2012 Sep;41(3):369-74.
- Bell CM, Pot SA, Dubielzig RR. Septic implantation syndrome in dogs and cats: a distinct pattern of endophthalmitis with lenticular abscess. Vet Ophthalmol. 2013 May;16(3):180-5.
- Verallo O, Fragiotta S, Verboschi F, et al. Diagnostic aspects and retinal imaging in ocular toxocariasis: a case report from Italy. Case Rep Med. 2012;2012:984512.
- Escanilla N1, Leiva M, Ordeix L, et al. Uveodermatologic lymphoma in two young related Portuguese water dogs. Vet Ophthalmol. 2012 Sep;15(5):345-50.
- Gallagher A. Leptospirosis in a dog with uveitis and presumed cholecystitis. J Am Anim Hosp Assoc. 2011 Nov-Dec;47(6):e162-7.
- Tetas Pont R, Matas Riera M, Newton R, et al. Corneal and anterior segment foreign body trauma in dogs: a review of 218 cases. Vet Ophthalmol. 2016 Sep;19(5):386-97.
- Denk N, Sandmeyer LS, Lim CC, et al. A retrospective study of the clinical, histological, and immunohistochemical manifestations of 5 dogs originally diagnosed histologically as necrotizing scleritis. Vet Ophthalmol. 2012 Mar;15(2):102-9.
- Lam DL, Axtelle J, Rath S, et al. A Rayleigh Scatter-Based Ocular Flare Analysis Meter for Flare Photometry of the Anterior Chamber. Transl Vis Sci Technol. 2015 Dec 11;4(6):7. eCollection 2015.
- Wasik B, Adkins E. Canine anterior uveitis. Compend Contin Educ Vet. 2010 Nov;32(11):E1.
- Wiggans KT, Vernau W, Lappin MR, et al. Diagnostic utility of aqueocentesis and aqueous humor analysis in dogs and cats with anterior uveitis. Vet Ophthalmol. 2014 May;17(3):212-20.
- Linn-Pearl RN, Powell RM, Newman HA, et al. Validity of aqueocentesis as a component of anterior uveitis investigation in dogs and cats. Vet Ophthalmol. 2015 Jul;18(4):326-34.
- Gilmour MA, Lehenbauer TW. Comparison of tepoxalin, carprofen, and meloxicam for reducing intraocular inflammation in dogs. Am J Vet Res. 2009 Jul;70(7):902-7.
- Gilmour MA, Payton ME. Comparison of the effects of IV administration of meloxicam, carprofen, and flunixin meglumine on prostaglandin E(2) concentration in aqueous humor of dogs with aqueocentesis-induced anterior uveitis. Am J Vet Res. 2012 May;73(5):698-703.
- Fischer CA. Granuloma formation associated with subconjunctival injection of a corticosteroid in dogs. J Am Vet Med Assoc. 1979 May 15;174(10):1086-8.